Trending News
This Is Why Everyone Needs To Prepare For The Day When Devastating Cyber Attacks Take U.S. Power Grids Down
(OPINION) What would you do if the power grid where you live went down and there was no...

Trending News
A section of the San Andreas Fault is beginning to wake up
Earthquakes are notoriously hard to predict, but one consistent earthquake in California—which...

Kirk Franklin draws backlash for attire at performance in Jamaican gospel concert
(OPINION) Gospel artist Kirk Franklin is drawing the ire of conservative Christians in Jamaica for...

Washington mother accused of stabbing 4-year-old son 41 times and dumping body on side of highway
A mother in the state of Washington is accused of stabbing her 4-year-old son 41 times and then...

DEVELOPING: Anti-Semitism growing in College Campuses across America
(OPINION) Imagine walking on your college campus or the campus your son or daughter attends and...

DEVELOPING: Israel preparing to advance ground operation into Rafah
Israel’s military is poised to evacuate Palestinian civilians from Rafah and assault Hamas...

UN nuclear chief warns that Iran is ‘weeks, not months’ from enough uranium to make a bomb
The head of the United Nations nuclear watchdog has said that it would take Iran just weeks to...

FALLING AWAY: Bible engagement falls below 40% among Americans, continuing a decade-long decline
(OPINION) A comprehensive survey conducted by the American Bible Society, “State of the...

Nearly 2,000 students and 15 teachers walk out of LGBTQ ‘Indoctrination’ lesson in Minnesota
Nearly 2,000 Minnesota high school students opted out of an “LGBTQIA+ History and...

Peter Schiff says get ready for ‘major dollar decline’ — predicts end of greenback as global reserve currency
(OPINION) Economist Peter Schiff warned Americans to prepare for “a major dollar decline.” The...

Anti-Israel agitators vow to stay on campuses until administrators cave to their demands
Students at a growing number of U.S. colleges and universities are forming anti-Israel protest...

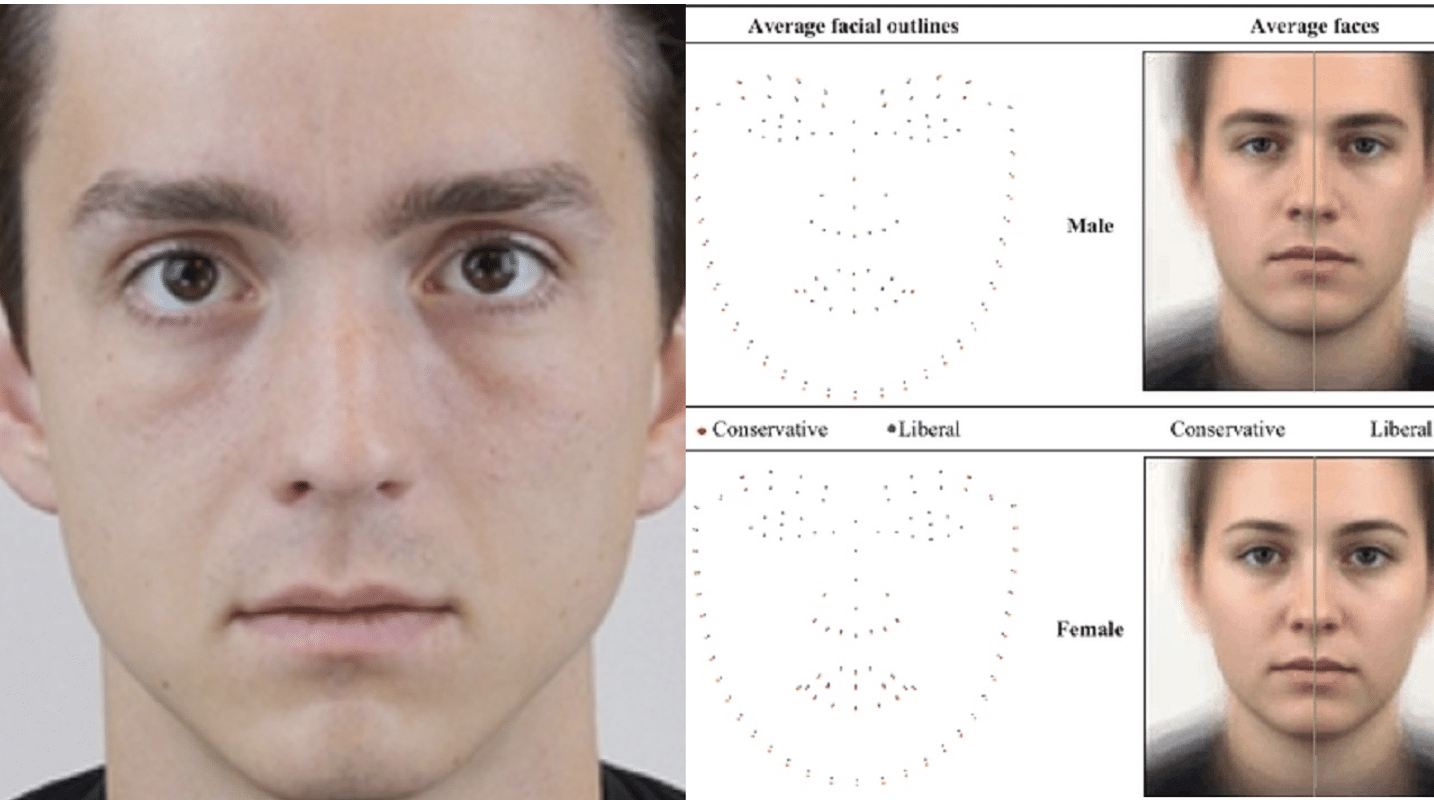
AI capable of predicting political orientations from blank faces
Researchers are warning that facial recognition technologies are “more threatening than...

Russian hackers remotely hack Texas drinking water
Russian hackers claiming to be backed by the Kremlin are believed to have remotely accessed a...