Trending News
Russian hackers remotely hack Texas drinking water
Russian hackers claiming to be backed by the Kremlin are believed to have remotely accessed a...

Trending News
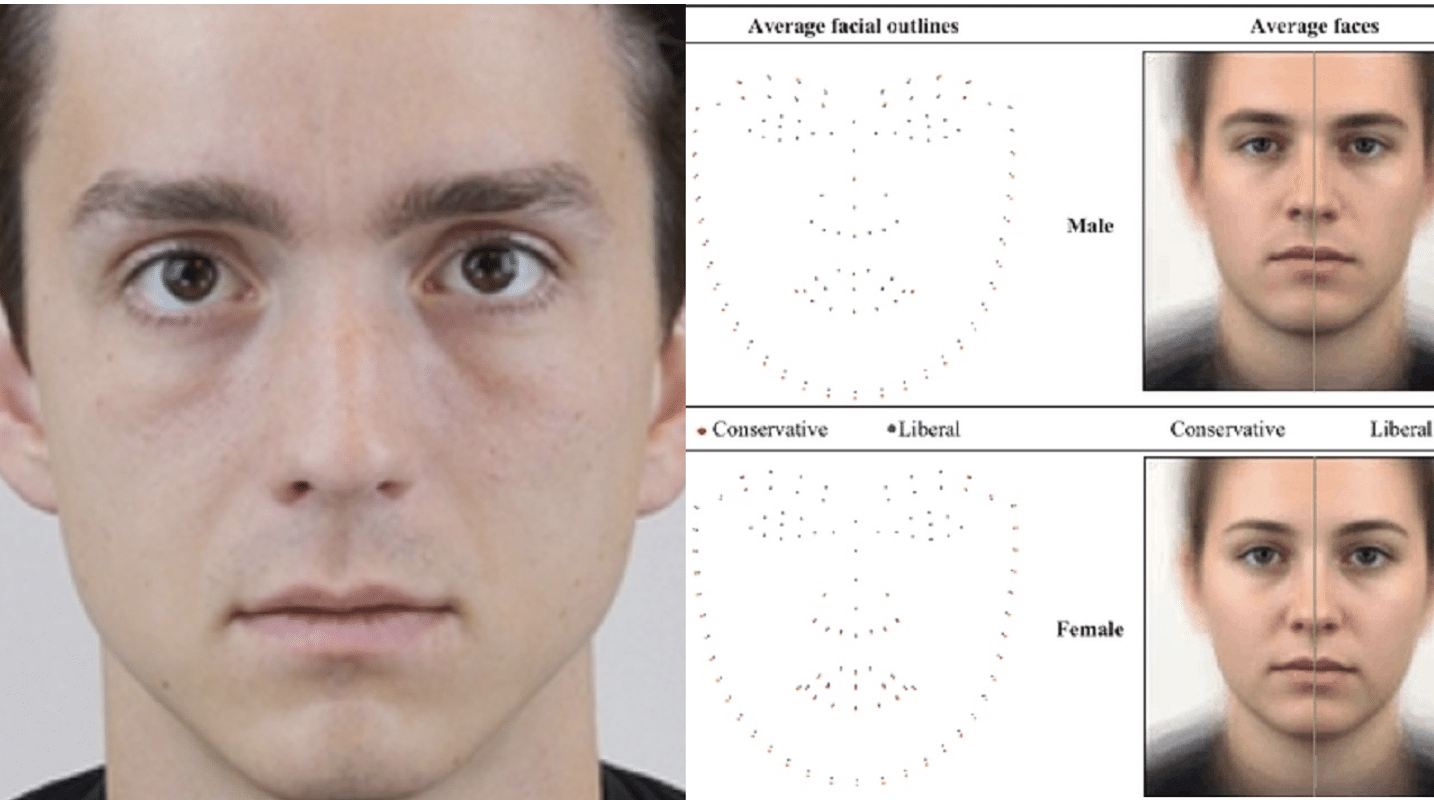
AI capable of predicting political orientations from blank faces
Researchers are warning that facial recognition technologies are “more threatening than...

(NEW PODCAST) Are They Giving Us A Warning About What Is Coming?
(OPINION) In tonight’s podcast, we discuss a recent event that transpired in the UAE,...

North Korea issues nuke ‘warning signal’ to America and South Korea
North Korea’s missile test on Monday was in fact a drill to test nuclear force preparedness,...

Taylor Swift’s new song lyrics are explicit, demonic and blasphemous but many Christians refuse to stop listening to her music
(OPINION) Are Taylor Swift’s lyrics full of “explicit” content? It’s one thing to drop a...

DRAMA: Megachurch Pastor condemns Mark Driscoll’s ‘Demonic’ Behavior, Apologizes for inviting sword-swallower to Conference
(OPINION) Yet another twist has emerged in the ongoing feud between outspoken Pastor Mark Driscoll...

Bridges collapsing and homes underwater as ‘once in a century’ floods threaten 127 million in China
At least three people have died with 11 still missing after severe floods only seen “once in...

Biden being urged to mobilize National Guard to protect Jewish college students
Sens. Josh Hawley and Tom Cotton called on President Biden to mobilize the National Guard to...

PROPHECY WATCH: Will the Nations ‘Impose’ a “Peace Agreement” upon Israel?
(OPINION) This last week, when Iran unleashed its barrage of 350 drones and missiles against the...

UNRAVELING: Anti-Israel agitators form human chain as police descend on NYU campus
Anti-Israel protesters on the campus of New York University formed a human chain Monday evening as...

Does it matter that Steven Furtick’s Easter sweater had a $2000 price tag?
(OPINION) The outfit Pastor Steven Furtick wore on Easter is still generating online conversation,...

(NEW PODCAST) What Passover Means For The New Testament Believer
(OPINION) In tonight’s podcast, we discuss the season of Passover and why it is significant...

America’s fight to save handwriting from extinction as IQs begin to fall for first time ever and teachers warn some 20-year-olds can’t sign checks anymore
Several US states are trying to prevent handwriting from going extinct as classrooms increasingly...