Trending News
DEVELOPING: Reports indicate Israeli strikes have taken place in Iran, Syria, and Iraq
An Israeli missile strike targeted a site in Iran early Friday morning, according to ABC News. The...

Trending News
Smart gun will require fingerprint and facial recognition technology to operate
A smart gun developed by Biofire will soon start shipping to customers who pre-ordered the...

Meteorologist warns of ‘Weather Wars’ between countries
Meteorologists have warned of potential “weather wars” between countries if cloud...

(NEW PODCAST) Number Of Black Eyed People Sightings Are Increasing
(OPINION) In tonight’s podcast, we discuss the alarming number of reports from eyewitness...

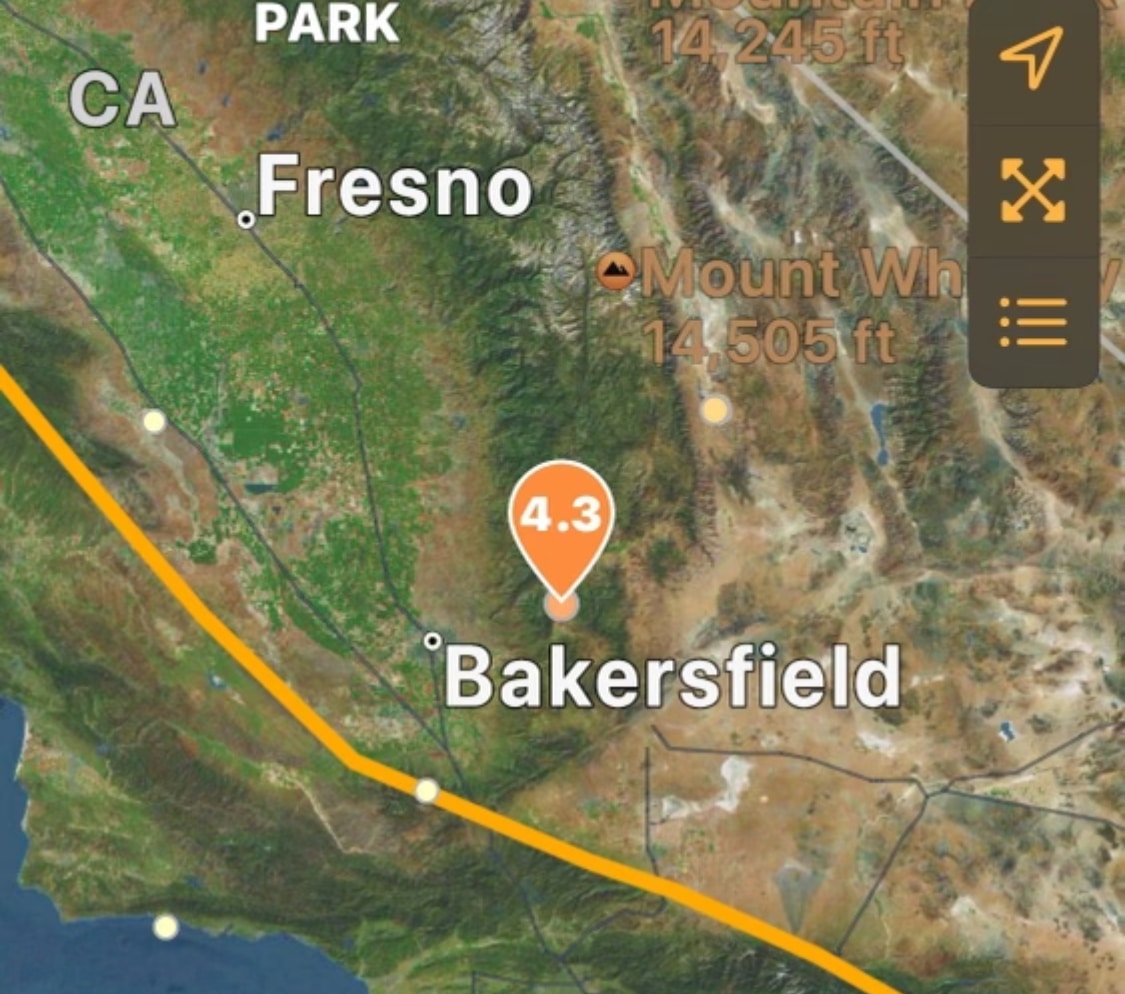
4.3 earthquake strikes near Bakersfield, CA
According to the United States Geological Survey, a 4.3 magnitude earthquake struck south of...

Two hunters ‘become first Americans to die from ZOMBIE DEER disease’ after eating infected meat
Two hunters may have become the first Americans to die from a ‘zombie deer’ disease....

Mystery flu spreading in Argentina ‘similar to Covid’ leaves over 60 people critically ill and doctors on “high alert”
In an outbreak that shares an eerie similarity with the arrival of Covid-19, a mystery illness has...

Middle School student drags fellow pupil to the ground and repeatedly bangs her head on the floor in front of crowd who are barking and cheering
This is the moment a student dragged a fellow classmate to the ground and repeatedly banged her...

Israel unlikely to strike Iran before the end of Passover
Israel appears unlikely to carry out a strike on Iran before the end of Passover, a US official...

Biden’s education secretary vows to shut down nation’s largest Christian university
After Department of Education Secretary Miguel Cardona vowed to shut down Grand Canyon University...

DEVELOPING: Widespread 911 Outages Are Reported in Four States
A large-scale outage is affecting residents’ ability to call the 911 emergency number in parts of...

Tsunami alert after a volcano in Indonesia has several big eruptions and thousands are told to leave
Indonesian authorities issued a tsunami alert Wednesday after eruptions at Ruang mountain sent ash...

John Lindell calls Mark Driscoll to repent for ‘Trying to destroy James River Church’ and ‘Sow disunity’ in the Lindell Family
(OPINION) During an evening service on Wednesday (April 17), Pastor John Lindell of James River...